At 1 year, 38% of the patients are progression-free, but the rest have progressed, meaning there’s got to be something better. Both at our main campus and throughout the care centers, that’s being used. But right now that has become the standard of care for the nonsquamous patient, and we’re using it at Yale Cancer Center, where I work. I think in the next months we’ll start seeing some more of those data emerge. We’re still trying to investigate exactly the biology of what’s happening when you give chemotherapy plus immune therapy. So now you do maintenance therapy and pemetrexed with pembrolizumab. The use of pemetrexed as a maintenance has been shown to be quite promising and active. So usually 4 cycles of chemotherapy, these are data from many years ago and that have continued to be held up 4 cycles of cytotoxic chemotherapy are enough. Chemotherapy is active and kills dividing cells, but there are dividing cells in the bone marrow, and in the hair, and in the lining of the gut. 
Well, you know, the human body can tolerate only so much chemotherapy. In those patients who are PD-L1 high, more than 50%, there would still be a discussion of trying to use the immunotherapy alone. So the data looked a little bit better for the PD-L1 high than the low and the 0, but the hazard ratios for survival were significant in all 3 groups.īased on that study for the most common type of lung cancer, non-small cell lung cancer, of which nonsquamous is the most common type of that, most people would use, after discussion with patients, pembrolizumab with carboplatin-pemetrexed. But importantly, it was also positive across all lines, at least in the first analysis that was presented at the AACR in 2018. And this trial, of course, was highly positive at its primary end point with hazard ratios in the 0.6 range. So carboplatin-pemetrexed and pembrolizumab. 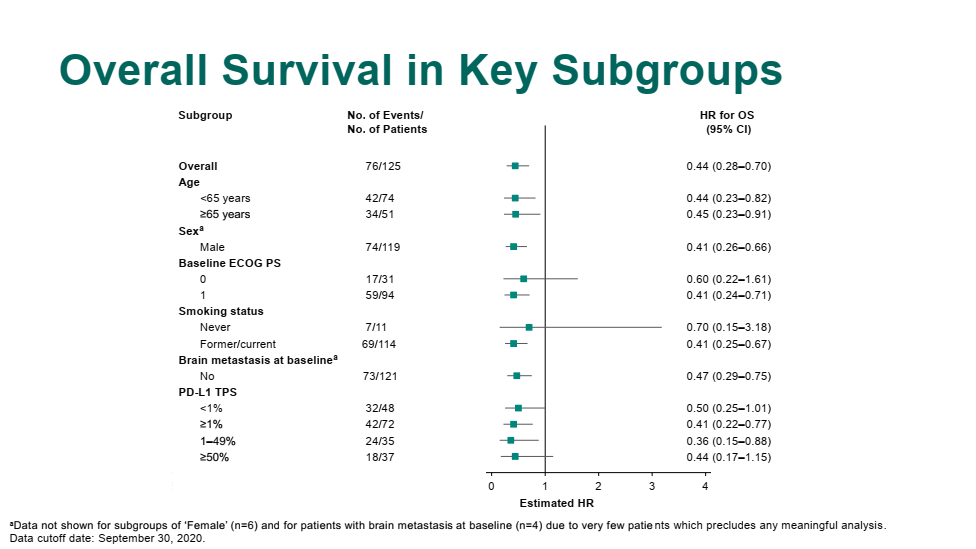
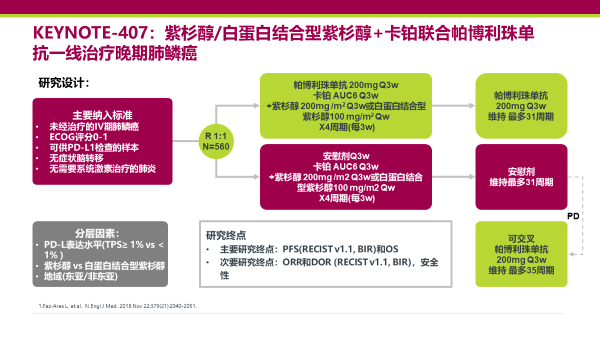
And the study takes patients, no matter what their PD-L1 status, and treats them with chemotherapy and carboplatin-pemetrexed because they’re all nonsquamous patients or chemotherapy with pemetrexed and pembrolizumab. But even there, the response rates are less than 50%, although the results are quite extraordinary. Roy Herbst, MD, PhD: KEYNOTE-189 is an important study because it took pembrolizumab, which we know can be used as a single agent in people who are PD-L1 high. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
